- Abstract
-
- Yawning was recorded from five rhesus
monkeys restrained in a chair after i.m.
injection of dopaminergic compounds: apomorphine
(0.03 mg/kg), quinpirole (0.01 mg/kg), and
(-)-3-(3-hydroxyphenyl)-N-(1-propyl)piperidine
(1 mg/kg). SKF 38393 or physostigmine produced
no yawning. Yawning from apomorphine was blocked
by chlorpromazine or SCH 23390 (0.03 mg/kg).
Sulpiride (10 mg/kg) was ineffective. The
difference between rats and monkeys in their
yawning response to dopaminergic compounds is
discussed.
-
- 1. Introduction
-
- Dopamine agonists, such as apomorphine,
produce yawning in rats and humans. The
pharmacology of this phenomenon in rats has been
extensively studied. The dose-response
relationship is biphasic, with the optimal dose
for yawning always below that to elicit
stereotypic behaviors and the order of potency,
among the agonists, correlates with that of
dopamine autoreceptor activation using
biochemical endpoints (Gower et al., 1984).
There is, however, no direct evidence relating
the yawning response to dopamine autoreceptor
stimulation. The time course of yawning after an
injection of apomorphine is also different from
that of the reduction of striatal extracellular
dopamine level as expected from dopamine
autoreceptor activation (Stahie and Ungerstedt,
1990).
-
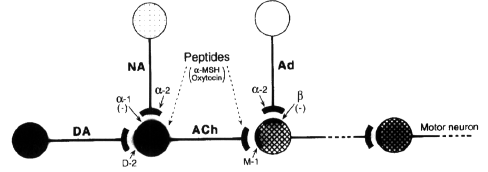
- The role of the dopamine D, receptor in the
yawning response to dopamine agonists in rats is
a matter of controversy. The D1-selective
partial agonist, SKF 38393, produces no yawning
when given alone. But, both antagonism
(Zarrindast and Poursoltan, 1989;) and
potentiation (Spina et al., 1989) by SKF 38393
on ciopantinergic yawning in rats have been
reported. Likewise, antagonism of
dopaminergic-induced yawning in rats by SCH
23390, a selective D1 antagonist, was found by
some investigators (Morelli et al., 1986), but
not others (Zarrindast and Poursoltan, 1989).
Mixed
-
- dopamine DJ/D, antagonists (neuroleptics)
are generally effective blockers of
dopaminergic-induced yawning in rats (Dubuc et
al., 1982).
-
- A limited study of dopaminergic-induced
yawning in human subjects indicates that it
occurs at sub-emetic doses (LaI, 1988). We are
not aware of any similar study in subhuman
primates. This study investigates
dopaminergic-induced yawning in rhesus
monkeys.
-
- 2. Materials and methods
-
- 2.1. Animals
-
- Each compound was studied in five adult
rhesus monkeys. All but one (monkey No. 659) are
males. The animals were selected for their calm
behavior when restrained in a chair and for
their yawning response to apomorphine in an
initial screening test. Between experimental
sessions, the monkeys were housed in individual
cages, with room lighting at a 06:00-18:00 h
daily cycle. They were fed approximately 120 g
of Purina Monkey Chow daily in the afternoon,
and had drinking water available at all times.
The animals were used once a week in the yawning
experiments.
-
- 2.2. Procedure
-
- During experimental sessions, the animals
were placed in a plexiglas primate restraining
chair (Plas Labs), which was housed in a
sound-attenuating cubicle with a 60-W light in
the ceiling and forced-air ventilation. A
speaker in the cubicle provided masking noise. A
one-way mirror window allowed video-recording of
the face of the animal during the experimental
session.
-
- In the morning of an experimental day, the
animal was taken from its home cage, placed in
the chair, and allowed 30 min for acclimation.
During this time, behavior was recorded. An i.m.
injection was then given, and recording
continued without interruption for an additional
40 min. For antagonism studies, the antagonist
was injected at the beginning of the 30-min
acclimation period.
-
- After the experiment, the videotape was
viewed, and the number of yawns and periods of
time when both eyelids were closed was recorded.
Yawning is distinguished from other mouth
movements by full extension of the jaws,
withdrawal of the lips, and exposure of
teeth.
-
- 2.3. Drugs
-
- The following drugs were used: apomorphine
HCI (Sigma), quinpirole HCI (Eli Lilly and Co.),
S-( - )-3(3-hydroxyphenyl)-N-( 1
-propyl)piperidine (S(- )-3PPP) HCI (Research
Biochem., Inc.), (± )-SKF 38393 HCI
(Research Biochem., Inc.), physostigmine
salicylate (Sigma), (+ )-SCH 23390 HCI (Research
Biochem., Inc.), (± )-sulpiride (Sigma),
and chlorpromazine HCI (Sigma). All the
compounds were dissolved in saline solution and
injected i.m.
-
- 3. Results
-
- Spontaneous yawning was evaluated by
following the experimental protocol and
injecting saline after the 30-min acclimation.
Each of the five monkeys was studied in four
different experimental sessions. The frequency
of spontaneous yawns did not exceed one yawn per
monkey per session. Spontaneous yawning was,
therefore, an extremely rare occurrence under
these experimental conditions. Likewise, there
were no significant periods of eyelid closure
during control sessions. The maximum response
was 23 s in 30 min for one monkey.
-
- Injections of apomorphine and quinpirole
produced yawning in all five monkeys, with a
significant change from saline at 0.03 and 0.01
mg/kg, respectively (table 1). The effect has a
fast onset, and seldom lasted more than 20 min.
(- )-3-PPP also produced yawning, but only at
much higher doses (P <0.05 at 1 mg/kg), and
did not reach the same frequency as did the
other two compounds. For monkeys B53, B57 and
659, a biphasic dose-response relation is noted
in (- )-3-PPP-induced yawning. SKF 38393 (3
mg/kg) produced no yawning. Pretreatment with 3
mg/kg of SKF 38393 30 min before a challenge
dose of quinpirole (0.003 mg/kg) increased
yawning in two of the five monkeys.
Physostigmine was tested at 0.03 mg/kg with no
yawning observed. A higner dose (0.1 mg/kg) of
physostigmine produced excessive salivation and
mouth movement, making evaluation of yawning
difficult.
-
- For each experimental session, the video
recording was also examined for periods of
eyelid closure as an indicator of drowsiness and
sleep. The dopamine agofists, at effective doses
for yawning, produced only brief periods of
sleep in two to three monkeys. During 20 min of
observation, the best responders had the
following cumulated time of eyelid closure:
apomorphine (81 s, 0.03 mg/kg, monkey 659),
quinpirole (146 s, 0.03 mg/kg, monkey 57B), and
(- )-3-PPP (4 s, 0.3 mg/kg, monkey B53).
Yawning, therefore, was not accompanied by any
significant sign of drowsiness.
-
- Several dopamine antagonists were tested for
blockade of the yawning response from
apomorphine (fig. 1). Both chlorpromazine (0.1
mg/kg) and SCH 23390 (0.03 mg/kg) antagonized
the yawning (P <0.05). The selective D2
antagonist, sulpiride, was ineffective at doses
up to 10 mg/kg. The combination of
chlorpromazine or SCH 23390 with apomorphine
resulted in increased eyelid closure in some,
but not all, monkeys. The addition of sulpiride
to apomorphine did not increase eyelid
closure.
-
- 4. Discussion
-
- We have shown in this study that rhesus
monkeys responded to dopamine agonists with
increased yawning, similar to that observed
in rats and humans. In contrast to rats
(Zarrindast and Poursoltan, 1989), no yawning
was observed in monkeys after physostigmine. The
effective doses for yawning were quite low when
compared to other behavioral responses from
dopaminergic stimulation. For instance, we have
studied the discriminative stimulus effect of
apomorphine in monkeys (Tang and Code,
[989). In that study, 0.01 and 0.03 mg/kg of
apomorphine or 1 mg/kg of ( - )-3PPP produced no
effect on the rate of lever-press for food
pellets.
-
- An absence of eyelid closure at the yawning
doses indicated no sign of drowsiness. We
previously used an identical procedure to
observe the sedative effect of benzodiazepines
by recording eyelid closure in monkeys. No
yawning was seen at doses of triazolam or
diazepam which produced prolonged eyelid
closure.
-
- The dopaminergic-induced yawning in monkeys,
therefore, appears to be unrelated to the
sedative effects observed with these compounds
in humans.
-
- (- )-3-PPP produced a significant increase
in instances of yawning in rhesus monkeys, but
not to the same frequency as apomorphine or
quinpirole. This compound has been reported to
produce a very low, statistically insignificant,
occurrence of yawning in rats (Gower et at.,
1984; Stable and Ungerstedt. 1984). Since (-
)-3-PPP produced yawning in human subjects at
i.m. doses of about 30 mg (C. Tamminga, personal
communication), the monkeys appear to be a
sensitive species, like humans, for this
response. The relatively low frequency of
response from (- )-3-PPP, however, is consistent
with this compound being a dopamine partial
agonist.
-
- It is surprising that in this study the
D,-selective antagonist, sulpiride, failed to
block the apomorphineinduced yawning, since
studies in rats reported complete blockade (e.g.
Dubuc et at., 1982). It is possible that the
30-min pretreatment time was too short for this
compound to reach optimal blood level. However,
we did find in an earlier study that the 10
mg/kg dose of sulpiride blocked the
discriminative cue effect of apomorphine (0.1
mg/kg) with a similar pretreatment protocol
(Tang and Code. 1989). It is possible that the
mechanism/site of the yawning response has a
greater D, receptor reserve in monkeys than in
rats, which requires a greater antagonist
concentration for complete blockade. A higher
dose of sulpiride is expected to be effective,
although the poor solubility has kept us from
testing such doses in this study.
-
- The effective antagonism of SCI-I 23390
confirms a permissive role of the D1 receptor in
the yawning response. This hypothesis is not
necessarily inconsistent with the fact that the
D1 agonist, SKF 38393, had no effect when given
alone and had only a modest effect potentiating
quinpirole. If a certain threshold level of D1
activation is all that is needed, additional D1
stimulation may have no greater effect. Since
SKF 38393 is a partial agonist for the receptor,
it may, in fact, behave as an antagonist for
that receptor under certain circumstances
(Zarrindast and Poursoltan, 1989). In summary,
the results in this study suggest that
dopaminergic-induced yawning involves both and D
receptors. The relative importance of the two
receptor subtypes depends on the species, with
the rhesus monkey perhaps being more similar to
humans than is the rat.
-
- Gessa GL
Stretchings and yawnings induced by
adrenocorticotrophic hormone
Nature 23/07/1966; n°5047;
p426-427
- Gessa GL
Stretching and yawning movements after
intracerebral injection of ACTH
Revue Canadienne Bioliologie;
1967, 26, 3, 229-236
- Hipolide DC, Lobo
LL, De Medeiros R, Neumann B, Tufik S
Treatment with dexamethasone alters yawning
behavior induced by cholinergic but not
dopaminergic agonist Physiol
Behav 1999 Jan 1-15;65(4-5):829-32
- Kimura H,
Yamada K, Nagashima M, Matsumoto S, Ishii Y,
Yoshida S, Fujii K, Furukawa T Role of
adrenergic neuronal activity in the yawning
induced by tacrine and NIK-247 in rats.
Pharmacol Biochem Behav 1992
Dec;43(4):985-91
- Kimura
H, Yamada K, Nagashima M, Furukawa T
Involvement of catecholamine receptor activities
in modulating the incidence of yawning in
rats.Pharmacol Biochem Behav
1996 Apr; 53(4):1017-21
- Laing J, Ogilvie R
EEG correlats of yawning during sleep onset
Sleep Research 1988; 17; p98
- Lobo LL,
Neumann BG, Eidman DS, Tufik S Effects of
REM sleep deprivation of ACTH-induced yawning.
Pharmacology
1990;40(3):174-8
- Molgilnicka
E REM sleep deprivation changes behavioral
response to catecholaminergic and serotoninergic
receptor activation in rats
Pharmacol Biochem Behav 1981;
15; 1; 149-151
- Stahle
L Do autoreceptors mediate dopamine
agonist-induced yawning and suppression of
exploration ? a critical review.
Psychopharmacology 1992; 106;
1-13
- Neumann BG,
Troncone LR, Braz S, Tufik S Modifications
on dopaminergic and cholinergic systems induced
by the water tank technique: analysis through
yawning behavior Arch Int
Pharmacodyn Ther 1990
Nov-Dec;308:32-8
- Stahle
L Do autoreceptors mediate dopamine
agonist-induced yawning and suppression of
exploration ? a critical review.
Psychopharmacology 1992; 106;
1-13
- Tufik
S Does REM sleep deprivation induce
subsensitivity of presynaptic dopamine or
postsynaptic acetylcholine receptors in the rat
brain? European Journal of
Pharrnacology 1987; 140; 215-219
- Tufik S
Effects of stress on drug induced yawning
Physiology & behavior 1995;
vol 58; n 1; p 1881-18
|



